- Review
- Open access
- Published:
Role of nutraceuticals during the transition period of dairy cows: a review
Journal of Animal Science and Biotechnology volume 11, Article number: 96 (2020)
Abstract
The transition period of dairy cattle is characterized by a number of metabolic, endocrine, physiologic, and immune adaptations, including the occurrence of negative energy balance, hypocalcemia, liver dysfunction, overt systemic inflammatory response, and oxidative stress status. The degree and length of time during which these systems remain out of balance could render cows more susceptible to disease, poor reproductive outcomes, and less efficient for milk production and quality. Studies on both monogastrics and ruminants have reported the health benefits of nutraceuticals (e.g. probiotics, prebiotics, dietary lipids, functional peptides, phytoextracts) beyond nutritional value, interacting at different levels of the animal’s physiology. From a physiological standpoint, it seems unrealistic to disregard any systemic inflammatory processes. However, an alternate approach is to modulate the inflammatory process per se and to resolve the systemic response as quickly as possible.
To this aim, a growing body of literature underscores the efficacy of nutraceuticals (active compounds) during the critical phase of the transition period. Supplementation of essential fatty acids throughout a 2-month period (i.e. a month before and a month after calving) successfully attenuates the inflammatory status with a quicker resolution of phenomenon. In this context, the inflammatory and immune response scenario has been recognized to be targeted by the beneficial effect of methyl donors, such as methionine and choline, directly and indirectly modulating such response with the increase of antioxidants GSH and taurine. Indirectly by the establishment of a healthy gastrointestinal tract, yeast and yeast-based products showed to modulate the immune response, mitigating negative effects associated with parturition stress and consequent disorders.
The use of phytoproducts has garnered high interest because of their wide range of actions on multiple tissue targets encompassing a series of antimicrobial, antiviral, antioxidant, immune-stimulating, rumen fermentation, and microbial modulation effects. In this review, we provide perspectives on investigations of regulating the immune responses and metabolism using several nutraceuticals in the periparturient cow.
Background
Around parturition, dairy cows experience the majority of health problems as a consequence of improper adaptations from a non-productive period (dry) to the onset of a new lactation. Thus, the purpose of many researches, throughout the second half of the XX century and current day, has been focused on the influence of the “transition period” (TP), to date established to start at the beginning of dry-off, on health and immune function, the interplay between the endocrine and immune systems, and, more recently, nutrition linked to immune function.
To a large extent, the health problems during the periparturient period relate to cows having difficulty in adapting to the nutrient needs for lactation [1]. This may result in physiological imbalance (Fig. 1), a situation where the regulatory mechanisms are insufficient for the animals to function optimally leading to a high risk of a complex of digestive, metabolic [3], and infectious problems [2]. The risk of infectious diseases increases if the immune functions (such as phagocytosis, oxidative burst, chemotaxis, cell-cell interaction) are impaired. Nutrition plays a pivotal role in immune response, and the effect of nutrition may occur directly through nutrients or indirectly through biological active metabolites, for example, in situations with physiological imbalance [1].
Theoretical pattern of changes in the main physiological aspects of healthy subjects during the transition period. Ideally, the Negative energy balance (NEB), inflammation, and oxidative stress would be close to zero (i.e. absence of the phenomena), whereas the immunocompetence and the calcemia would be close to 100% of their optimal level [2]
Use of nutraceuticals has received increasing attention for the improvement of animal health, welfare, and productivity in herd health management. Studies on nutraceuticals investigated their use as nutrients, dietary supplements, herbal products, and processed feeds (including dietary fiber, probiotics and prebiotics, polyunsaturated fatty acids, antioxidant vitamins and trace minerals, and phytoactive compounds). Results revealed their potential to support the immune system and metabolic activity of the main organs (such as liver, mammary gland, and gut) prior to and after parturition, especially when inflammatory response activate various components of the immune system and alterations in metabolism [4]. These motives have promoted the value of nutraceuticals, where complex multitarget poly-pharmacological mechanisms, such as activation of antioxidant defense and anti-inflammatory pathways (along with beneficial effects on cells through integrity, survival, proliferation, and differentiation), are exhibited [5].
Considering the present framework, this review aims to summarize findings and potential perspectives of those nutraceutical compounds that are not practically considered in the diet requirement formulations for high-yield dairy cows, especially during the transition period. We also know the relevant importance of certain vitamins and minerals functioning as nutraceuticals to modulate the oxidative stress response. However, in this review, we do not specifically discuss their use and outcomes since several scientific impacting and influential reviews have been already published [6].
The transition cow – an overview
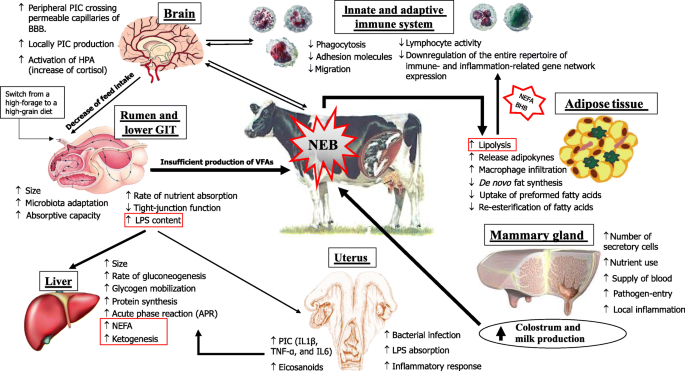
In one of his influential review articles published in 1999, Prof. J. Drackley [7] argued that the biology underlying the transition to lactation was the “final frontier” in our understanding of the dairy cow. Since then, a number of relevant in-depth studies have uncovered most of the “obscured field” of the transition period. Such researches have demonstrated that immune cells are directly involved in a surprising array of metabolic functions, including the maintenance of gastrointestinal function, control of adipose tissue lipolysis, which in turn determines the liver functionality, and regulation of insulin sensitivity in multiple tissues [8]. On the other hand, it was also postulated and highlighted that metabolic changes related to energy and calcium supply in support of lactation, occurring concurrently, impair the innate immune response [9, 10] (Fig. 1). Clearly, the mechanisms linking these changes and metabolic challenges during the transition period (Fig. 2) are only partially understood, further demanding the question: what is the trigger of the metabolism and immune imbalance in the peripartum?
Peripartal adaptations of the key tissue such as liver, mammary gland, adipose, rumen, uterus, brain, as well as the immune system. It is highlighted the main factors that influence the functional response capacity of the key tissues involved in the homeorhetic adaptation during the transition period. NEB: negative energy balance; GIT: gastro-intestinal tract; BHB: β-hydroxybutyrate; NEFA: Non-esterified fatty acids; LPS: lipopolysaccharide; HPA: hypothalamus-pituitary-adrenal axis; PIC: pro-inflammatory cytokines; BBB: blood-brain barrier
During TP and mainly after parturition, circulating glucose is prioritized to the non-insulin-dependent glucose transporters, which are only expressed on immune cells and the mammary gland. Nevertheless, the massive glucose requirements of an activated immune system during systemic inflammation could further reduce the energy available for the mammary gland, aggravating the negative energy balance (NEB) occurring in early lactation [11]. Data obtained in Holstein lactating cows after a stimulation with LPS indicated that an acutely activated immune system uses > 1 kg of glucose within 720 min from the onset of inflammation [11]. When NEB occurs, mobilization of body fats and proteins are induced, and non-esterified fatty acids (NEFA) and amino acids are used as fuel sources by the liver [12]. However, a severe NEB occurring in TP could induce a NEFA overload in the liver, increasing the release of beta hydroxybutyrate (BHB) in blood and impairing pivotal functions [7].
Oxidative stress also occurs during this period and is driven by the imbalance between the production of reactive oxygen metabolites (ROM), reactive nitrogen species (RNS), and the neutralizing capacity of antioxidant mechanisms in tissues and blood. The increase in oxidative stress and inflammation during this period is also associated with a reduction in liver functionality, for which measurement of APP can provide a useful tool to assess liver function as well as inflammation [13]. In this context, it is particularly important to understand how inflammatory processes in the peripheral tissues of transition cows signal their anorexic action to the hypothalamus [14]. Indeed, the hypothalamus is one of the key regions of the brain regulating energy balance as it receives and integrates input signals from the periphery, sensing humoral substances (such as nutrient-related metabolites, hormones and cytokines), but also integrating neural signals from other brain regions, the tongue or oronasal origin to adjust feed intake and energy expenditure [15] (Fig. 2).
Herein, we would like to point out the scenario occurring in the rumen during the transition period. Few studies have investigated the molecular adaptations of ruminal epithelium during the peripartum period [16]. These studies revealed the existence of interactions among genes of the immune system and those involved in the preparation for the onset of lactation, as well as the presence of growth factors that seem to be regulated after parturition [17]. The connections among ruminal fermentation, the ensuing ruminal epithelium adaptations, and the consequent system responses (Fig. 2) of the cow remain unclear. However, whether microbial metabolism could affect epithelial gene expression via metabolites remains uncertain. Also, the interaction of rumen epithelium with systemic immune response opens a new scenario in the management of forestomaches. The role of diet appears to be crucial (e.g. fermentability of carbohydrates, protein degradability) for nutrient balance and/or for microbiota composition, which might alter epithelium functioning (e.g. increase its permeability). Thus, feed molecules that favor rumen stability should be studied to discover their effects on modulation of the rumen fermentation, microbiota biodiversity, and protection of epithelial cells.
Essential fatty acids
Mammals are able to synthetize all fatty acids that are essential for normal physiologic functions, except for polyunsaturated fatty acids (PUFA) ascribed in the n-3 and n-6 family, or the so-called “essential fatty acids,” as they must be provided through the diet. In TP, the amounts of PUFA decrease substantially in all the body compartments as compared with mid-lactation cows [18, 19], while the proportion of several saturated fatty acids (SFAs) increases [7, 20]. The primary and major source of n-3 fatty acids in ruminants is forage, particularly for grazing cattle, since forage galactolipids are rich in α-linolenic acid (ALA; C18:3n-3). Supplemental sources of n-3 PUFA in dairy cows diets include ALA from flaxseed as well as eicosapentaenoic (EPA; C20:5n-3) and docosahexaenoic acids (DHA; C22:6n-3) from fish oil [21]. Conversely, the n-6 PUFA are contained in many different feedstuffs (i.e. soybean, sunflower, corn, and cottonseeds), and their intake substantially increases around and after calving.
PUFAs
Supplementing PUFAs in dairy cows’ diets differentially affects productive performances depending on the dose and type administered (Table 1). Fish oil is known to decrease feed intake when supplemented in non-rumen protected forms and when percentages included in the diet are higher than 1% DM [21]. Decreased DMI reported with PUFAs could account for the reduced milk yield (MY) reported in several experiments. Furthermore, reduced butterfat has also been reported in several studies, administering PUFAs in different phases and suggesting incomplete protection against rumen biohydrogenation to intermediate isomers that are known to depress milk fat or are associated with milk fat depression (e.g. trans-10,cis-12 conjugated linoleic acid) [34]. Nevertheless, such results on productive performances are not consistent throughout the studies, indicating the pivotal role of diet formulation and lactation phase in affecting PUFAs’ effects on MY and composition.
Supplementing rumen protected PUFAs during TP reduces the proportion of circulating SFAs, which markedly increase in the blood of early lactating cows due to the massive mobilization of NEFA from adipose tissue [20]. Such a shifted profile of circulating fatty acids is reflected in other body compartments. Increasing the amount of PUFAs in oocytes and follicular fluid has shown a positive effect on embryo implantation (EI), as excessive amounts of SFAs are known to impair oocyte competence and development [21]. A higher inclusion of PUFAs in white blood cells’ membranes at the expense of SFAs positively affect the immune functions [35]. In fact, excessive inclusion of SFAs resulting from adipose tissue mobilization in leukocytes membrane is known to play a role in triggering immune dysfunctions and unregulated inflammation in early lactation [36]. High amounts of SFAs modify proteins through fatty acylation, altering membrane fluidity, influencing how proteins anchor to the plasma membrane, and affecting the formation of glycoproteins that compose lipid rafts involved in lymphocytes activation, antibodies production, and inflammation [37].
Several SFAs (i.e. C12:0, C14:0 and C16:0) could also induce inflammation as they are similar to the acyl chains composing lipid A associated with bacterial lipopolysaccharides, which activate nuclear factor-κβ (NF-κB) mediated gene expression and increase inflammation and respiratory burst activity [19, 38]. Supplementing essential PUFAs (as rumen protected mainly) also exerts a direct effect on immune cells through modulating the expression of several transcription factors exerting pro- or anti-inflammatory actions. All n-3 PUFAs down-regulate the expression of adhesion molecules involved in inflammatory interactions between leukocytes and endothelial cells [39]. Linoleic acid (LLA; C18:2n-6), and particularly its conjugated isomers cis-9,trans-11 and trans-10,cis-12, interacts with peroxisome proliferation activated receptor (PPAR)-γ, while long chain n-3 (EPA and DHA) interacts with Toll-like receptors (TLRs)-2 and 4, PPARs, and sterol response element binding protein family [40, 41]. All these genes are involved in NF-κB regulation that orchestrates the production of pro-inflammatory cytokines in both immune and nonimmune cells [42]. The lack of n-3 and n-6 PUFAs in the post-partal period could, thus, induce uncontrolled inflammation.
n-6:n-3 ratio
The n-6:n-3 is the main indicator of PUFA, where values between 3.9 to 5.9 in dairy cows rations have been related to positive effects on immune functions and reproductive performances [43], while higher values are associated to the massive use of n-6 sources. Supplementing n-3 sources affects the composition of plasma, reducing the n-6:n-3 ratio to between − 3.7 to − 40.7 [26, 29]. Such a shift of fatty acids composition is reflected in many body compartments (i.e. cell membranes, follicular fluid, granulosa cells and oocytes), exerting different effects based on the dose, physical form, and administration time of n-3 rich feeds [21].
Decreased amount of arachidonic acid (AA; C20:4n-6) in cell membranes and follicular fluid induces anti-inflammatory and pro-resolving effects through shifting the oxylipids profile, class of lipid mediators produced by the enzymatic and non-enzymatic oxidation of PUFA [44], in favor of resolvins, protectins, lipoxins, and the 3 series prostaglandins and reducing the production of series 1 and 2 prostaglandins [45, 46]. This shifted oxylipid profile also improves the function of leukocytes (Table 2), as prostaglandin E2 is known to decrease the production of interferon gamma and proliferation of lymphocytes [65]. Altered oxylipid profile also improves the EI through ameliorating the development and maturation of oocytes [66, 67]. In fact, the series 1 and 2 prostaglandins (especially PGF2α released from the endometrium) are intimately involved in uterine involution and subsequent ovulation post-partum, negatively affecting the development of the morula and blastocyst stages [68]. Conversely, the 3 series prostaglandins improve the environment for embryo implantation and survival [64], and their production increases the lifespan of the corpus luteum, improves blastocyst cell numbers, and improves the maintenance of pregnancy [69]. Reduction in the concentration of linoleic acid (LA; C18:2n-6) in the follicular fluid also contributes to improving EI. In fact, AA is known to up-regulate the expression of steroidogenic acute regulatory protein, which mediates the transfer of cholesterol from the cytosol to the inner mitochondrial membrane, playing a pivotal role in steroid synthesis [70, 71].
Thus, high concentration of AA in the follicular fluid increases the production of estradiol, impairing oocytes implantation. An indirect effect driven by altered concentrations of hormones and metabolites in the follicular fluid surrounding the oocytes has also been hypothesized to concur in ameliorated EI driven by n-3 PUFA [66]. While supplementing n-3 PUFA has shown to improve the dynamics of follicular development (FD) in the ovary (Table 2), the mechanisms controlling such effects are still unknown [21]. Finally, supplementing n-3 PUFA is also known to improve embryo survival (ES) in late pregnancy (Table 2), as n-3 PUFA is essential for developing the central nervous system and reproductive system in unborn calves [56,57,58].
Methyl donor supplementation
Choline
Choline (beta-hydroxyethyltrimethylammonium hydroxide), an essential nutrient with various functions, is an essential component of various membrane phospholipids (phosphatidylcholine, lysophosphatidylcholine, choline plasmalogen, and sphingomyelin), a precursor for the synthesis of the neurotransmitter acetylcholine, and a source of labile methyl groups [72]. Besides, it is involved in lipid metabolism as a lipotropic agent. In fact, it is also a component of very low-density lipoproteins (VLDL) that carry fatty acids from the liver to peripheral tissues, thus playing a pivotal role in preventing fatty liver syndrome. Its need is strictly related to the other components of the 1-carbon metabolism, such as methionine, betaine, folic acid, and vitamin B12. The latter represents an important set of reactions involved in the synthesis of lipids, proteins, nucleotides, antioxidants, and methylation reactions [73].
Although choline is contained in various dietary feedstuffs, such as soybean, cottonseed, sunflower, and rapeseed [74], it is extensively degraded in the rumen [75], resulting in a low intestinal absorption from dietary sources. Apart from the diet, another possible source of choline is represented by the de novo synthesis of phosphatidylcholine through sequential methylation of phosphatidylethanolamine with S-adenosylmethionine (SAM) as the methyl donor [72]. Methyl groups can also be synthesized de novo by the tetrahydrofolate (THF) system [72]. Since both are interchangeable as methyl donors, choline is considered an essential nutrient for mammals when there is a lack of methionine and folates [76]. Around calving, the endogenous synthesis does not always satisfy the cow’s needs, considering the high output of methylated compounds through milk, low input from the diet, and possible short supply of methionine. Additionally, when choline is lacking, the methyl group metabolism is conservative, with a low rate of catabolism and a high rate of de novo synthesis via the THF system. Consequently, choline can be seen as a limiting factor in early lactating cows [76].
It has been suggested that supplementation of rumen-protected choline (RPC) can affect performances (Table 3), but the results are not always consistent. The increase in DMI due to RPC supply may explain the effects on milk production. For instance, the function of choline as a lipotropic agent can have positive effects on butterfat production, as it improves lipid metabolism, increasing the VLDL synthesis, availability of FA for the mammary gland, and its incorporation into phospholipid membranes around fat globules. The composition of milk fatty acids can be influenced by the increased availability of preformed fatty acids via VLDL, as well as the milk protein yield, mainly because choline serves as methyl source sparing methionine [85]. The main effects of RPC supplementation are indeed on liver function (Table 4), reducing fat deposition in the liver. To a greater extent, the biosynthesis of phosphatidylcholine (PC) comes from the CDP-choline pathway (which involves choline in the first step). In turn, PC is responsible for hepatic lipoprotein assembly and secretion into bloodstream as, mainly, VLDL. Thus, choline improves VLDL synthesis through NEFA esterification into TAG, contributing to the decrease of NEFA load in the liver [95]. The latter leads to reduction of both BHB levels and fatty liver incidence, which is responsible for impaired gluconeogenesis [96]. Moreover, positive effects were detected on the immune system, where abundant antioxidant content and reduced oxidative stress status were observed in polymorphonuclear leukocytes (PMNL) and monocytes phagocytosis associated with RPC supplementation improved in the periparturient period [90].
Methionine
Methionine, a limiting amino acid in dairy cows [97] essential for milk protein synthesis, is involved in cysteine, glutathione, and taurine synthesis [98] and plays a central role in 1-carbon metabolism [73]. Considering that methionine and choline share similar fate [99], they also exhibit many common effects, such as the role in lipoprotein synthesis and as a methyl donor. In these pathways, SAM, which is synthesized from methionine, can be used as a precursor for phosphatidylcholine and homocysteine. Homocysteine, an intermediate of the 1-carbon metabolism, can enter the transsulfuration pathway, through which cysteine can be synthesized. Cysteine is a precursor of taurine and glutathione, two important antioxidants. The NRC [100] proposed a daily methionine requirement of 2.4% of metabolizable protein for lactating cows; however, diets frequently do not meet this need. Therefore, supplementation in rumen-protected form is needed because, like choline, methionine is degraded in the rumen.
Rumen-protected methionine supplementation in the first 2 weeks after calving enhances methionine serum concentrations, improving its availability, as demonstrated by Dalbach et al. [101]. Methionine can be supplemented as hydroxy-analog or in physically encapsulated form. Milk yield and butterfat can be positively affected by methionine supplementation (Table 3), even though the main effects have been observed in protein yield due to improved amino acid requirements [102]. The effects on milk yield and fat could be related to the enhanced availability of nutrients because of the positive effect on DMI. In fact, methionine supply helps in maintaining constant rates of DMI prepartum and in increasing DMI in early lactation. This result is consistent in many studies, which may be due to improved inflammatory status, reduced oxidative stress, and enhanced liver function related to methionine supplementation [81]. The improvement in these functions suggests that high-producing dairy cows adapt successfully to the new lactation and overall to the transition period following methionine supplementation.
Methionine supplementation may also have positive implications on immune cell function (Table 4) with increased phagocytosis from neutrophils, improved oxidative burst capacity, greater T-lymphocyte proliferation, and blood neutrophil-killing capacity [90, 92, 103]. Late pregnancy supply of methionine may have positive implications on calf development (Table 3) with greater calf body weight at birth and in the first weeks of life. Proper maternal supply could contribute a greater amount of available nutrients for the fetus and also improved colostrum quality, resulting in better inflammatory status and innate immune response [104].
Live yeast and yeast-based products
There is a growing interest from livestock producers to find alternatives to antibiotics and antimicrobials for the enhancement of growth performance, general animal health, and well-being [105]. Currently, the most utilized and studied approaches are live yeast and yeast-based products derived from the strain Saccharomyces cerevisiae.
Effect of live yeast and yeast-based products on rumen activity modulation and performances
In ruminant nutrition, strains of this eukaryotic microbe help to stabilize ruminal pH and to activate fiber-degrading bacteria in the rumen, leading to improved fiber digestibility [106]. Several works have indicated a higher abundance of lactate-using bacteria (e.g. Megasphaera and Selenomonas), thus confirming the role of yeast in decreasing lactic acid concentration and helping maintain normal ruminal pH. Yeast supplementation also increased the relative abundance of fibrolytic-degrading bacteria, such as Fibrobacter and Ruminococcus, which would enhance fiber digestion in the rumen. These findings are in accordance with observations reported more recently by Uyeno et al. [107], who determined that supplementing mid-lactation dairy cows with 10 g of active yeast cells on a daily basis for 21 days activated fibrolytic bacteria in the rumen.
Regarding performance outcomes (Table 5), several studies have reported positive effects, such as increased DMI [111] and milk production [116, 117] when cows were fed Saccharomyces cerevisiae fermentation products, while other indicated that supplementation did not affect DMI or milk yield [118, 119]. In their meta-analysis, Poppy et al. [120] showed that supplementation with Saccharomyces cerevisiae fermentation products increased DMI of dairy cows in early lactation (< 70 days after calving). However, interaction effects between fermentability of the basal diet and yeast supplementation on DMI were not addressed. Recently, in a metabolic study on beef heifers, Shen et al. [121] found that supplementation of a high-grain diet (52.8% starch) with Saccharomyces cerevisiae fermentation products elevated the ruminal minimum pH and reduced the duration of pH < 5.6 by 6 h compared with control heifers. The study also reported improved ruminal and total-tract NDF digestibility, suggesting that the negative effects of feeding a high-starch diet can be attenuated by supplementing with yeast fermentation products. Similarly, Shi et al. [122] investigated the effects of supplementing with yeast fermentation products on milk production and DMI. The authors observed a transiently increased DMI on days 1 and 5 after calving followed by an increased feed efficiency during the post-fresh period. Overall, researchers have suggested that yeast cultures may cause a number of effects in the rumen, including increased pH, numbers of cellulolytic bacteria, and rate or extent of ruminal fiber digestion and altered VFA concentrations. On the basis of these previous results, yeast cultures may increase fiber digestion, which could increase the rate of passage and, therefore, improve DMI [111, 123].
Supplementation with yeast fermentation products has shown to decrease milk urea nitrogen concentration in cows fed high-starch diet. Comparatively, supplementing cows yeast fermentation products led to higher blood glucose and lower BHB concentration at 42 d after calving, suggesting a greater energy supply from the diet. Conversely, Nocek et al. [113] found an increase of milk, fat-corrected milk, and energy-corrected milk in early lactation cows supplemented with a yeast culture and yeast culture plus enzymatically hydrolyzed yeast.
Effect of live yeast and yeast-based products on mucosal and systemic immunity, and metabolic response
Little is known regarding the direct and indirect positive effects on the immune system and its subsequent biomarkers. Thereby, mitigation of negative effects associated with metabolic stresses and disease remains limited as well (Tables 5 and 6). Such responses could be attributed to improved energy status due to the effects on digestive function or to activation of the immune system through sensing of yeast components in the gut and subsequent cross talk between immune cells. However, the exact mechanism is unclear, especially stressing the potential of the yeast response to influence mucosal immunity.
Accordingly, Chen et al. [125] detected a higher expression of TLR receptors in ruminal epithelium of steers classified as acidosis resistant compared with those classified as susceptible. On the other hand, mechanisms of endotoxin (LPS) tolerance are present in intestinal epithelial cells and act to avoid deleterious TLR activation by a toll-interacting protein [126]. Minuti et al. [16] concluded that the ruminal epithelium during the transition period most likely adapts to an increase in rumen LPS content due to higher diet fermentability, resulting in greater VFA production and suboptimal pH driving greater bacterial lysis. Feeding live yeast results in greater abundance of TLR4 expression in ruminal epithelium, leading to a quicker response by innate and adaptive immunity [124]. On the other hand, further studies need to be conducted in order to highlight the mechanism behind the interaction between supplemented yeast, rumen epithelium, and innate immunity, allowing to define as positive the better degree of activation to the shorter time of resolution of a certain inflammatory response.
In response to an acute inflammatory stress induced by lipopolysaccharide, Fink et al. [127] reported that yeast products improved the health of beef cattle during the receiving period (i.e. the first 50 days in the feedlot) upon arrival to a feedlot. Duff and Galyean [128] also reported that improvements in health (i.e. decreased morbidity) may be observed in yeast-supplemented cattle exposed to stress, such as that associated with bovine respiratory disease or bovine viral diarrhea. Sanchez et al. [129] reported that yeast cell wall supplementation in receiving cattle enhanced the metabolic response to an acute immune challenge (i.e. lipopolysaccharide), thus improving the probability of recovery and enhanced efficiency of incoming cattle. Specifically, the authors [129] reported that energy metabolism and nutrient utilization may have been enhanced in yeast supplemented heifers prior to an immune challenge characterized by increased insulin, and decreased NEFA accompanied by alterations in blood urea nitrogen.
Effects of supplementing live yeast or yeast fermentation products during the transition period are inconsistent if we look at the inflammation and immune response (Table 5). However, Knoblock et al. [115] reported a marked reduction of haptoglobin 7 days after calving in cows supplemented with a Saccharomyces cerevisiae fermentation product, indicating that reduced inflammation was likely achieved by increased feed intake after calving. It is right to report that these kinds of commercial yeast-products contain multiple vitamins and antioxidants (i.e. polyphenols), bioactive compounds, including fermentation end products, β-glucans, and other components of the yeast cell, making difficult to discern the specific role of yeasts. Thus, it is difficult to evaluate which specific or in-combination compounds modulate the immune response reported in both human and animal studies. For example, a key constituent of yeast cell walls, β-glucan, has been shown to enhance defense against infections in rodent models [130]. β-glucans are not efficiently absorbed, but they interplay with gut-associated lymphoid tissues, which, in turn, could substantially influence circulating immune cells [106]. These interactions provide a mechanism, whereby signals at the mucosal surface (such as recognition of antigens and release of cytokines) of the gastrointestinal tract can broadly affect the function of leukocytes (macrophages, neutrophils, and lymphocytes) that migrate to damaged or infected tissues [131]. Recently, Yuan et al. [132] found that a product containing yeast culture plus enzymatically hydrolyzed yeast at the rate of 0, 30, 60, or 90 g/d from 21 d before to 42 d after calving linearly increased plasma anti-ovalbumin IgG levels following 3 ovalbumin challenges, which, in turn, indicates enhanced humoral immunity. The authors also concluded that supplementation with a product containing yeast culture plus enzymatically hydrolyzed yeast enhanced measures of humoral and mucosal immunity and modulated uterine inflammatory signals and mammary gland health in transition dairy cows. This further suggests that the immune system could be better alerted if immunogenic stimuli occur.
Phytoproducts
Phytochemical or phytoextracts are bioactive compounds naturally present in plants and products of secondary metabolism. These have raised great interest from the scientific community from in vitro studies to in vivo application to prevent clinical conditions and improve performances. These compounds, at the chemical level, can be categorized into two major categories: carotenoids and polyphenol. Polyphenols (PP) comprise about 5000 different molecules that are sub-divided into 5 classes: phenolic acids, flavonoids, lignans, and stilbenes. Some of the more common plants containing polyphenols, notable for their curative properties, include thyme and oregano (thymol), clove (eugenol), juniper (pinene), dill (limonene), cinnamon (cinnamaldehyde), hot peppers (capsaicin), tea tree (terpinene), garlic (allicin), and anise (anethol) [133]. Other plants of interest are aloe, yerba mate, pomegrate, sylimarin, green tea, and Hottunya cordata. Phytochemicals have been investigated as feed additivities for their potential use as antioxidants, antimicrobiotics [134], and immune stimulators or modulators of rumen fermentation [135, 136] to improve the general welfare status (Table 7), metabolism (Table 8), and reduce antibiotic use [105]. These products can affect animals by modulating appetite or digestive functions and processes (e.g. fiber digestibility, level of volatile fatty acids production in rumen), interacting with immune, endocrine or metabolic systems, and increasing their performance (milk yield and composition, fertility) [145, 146].
Implementation these substances during the transition period of dairy cows could improve rumen functions that control ruminal pH and prevent sub-acute acidosis conditions, increasing duodenal flow of protein and reducing methane production and energy losses [147, 148].
Phenols are resistant to rumen microbial degradation and, thus, could reach the small intestine [142]. This potential could be translated into improved digestibility, microbicidal activity against pathogens, and better antioxidant status and immune response, which compensate the negative effect of negative energy balance (NEB) that characterizes the transition period. Concerning the method of administration, these compounds could be obtained from different plant parts (peel, seed, leaves or stem) and added into TMR as powder or essential oil (EO) form. The dosage of administration depends on the plant species, chemical compound, and concentration, while either a single extract or blend can be used for administration. Most phytoextracts are applied in the form of essential oils that are recognized to have antimicrobial activity. In particular, their beneficial effects occur at the bacterial membrane level by changing the structure and fluidity [149], inhibition of enzymes [150] and proteins, RNA and DNA of the cells [151], or altering the flow transmembrane cations [150]. Although both gram-positive and gram-negative bacteria are affected by EO activity [152], gram-negative are less sensitive [153,154,155], which could affect their potency and selectivity on rumen microbiota modulation mainly to reduce methane losses [137]. Drong et al. [137, 156] evaluated the effect of a commercial EO-mix (Tables 7 and 8) on performance, energy metabolism, and immunological parameters of cows during the transition period. The results showed no effect of the EO-blend on energy status or milk production and decreased dry matter intake, which may have been affected by the experimental design and dose administration. Benchaar et al. [138] and Tassoul et al. [133] also reported decreased dry matter intake without influencing nutrient digestibility, ruminal fermentation, and milk production using an EO-blend containing thymol, eugenol, vanillin, and limonene. Comparatively, Oh et al. [142] reported an immune-stimulatory effect due to activation and induction of the expansion of CD4 cells using an EO-mix (garlic, capsicum oleoresin) pulsed into the rumen. Braun et al. [141] highlighted the potential of EO to activate cation-transporting proteins, increasing the uptake of cations like calcium and ammonium at the ruminal epithelial level; in particular, calcium uptake plays an important role in the milk fever metabolic disorder. Yang et al. [143] suggested instead that garlic or juniper berry EO increased dry and organic matter digestibility correlated with increased crude protein rumen digestibility.
Tannins, a type of water-soluble polyphenolic compounds in plants, are considered a natural antioxidant due to the capability of their aromatic rings to combine with free radicals and form stabilized phenoxyl radicals [157]. Tannins and saponins have also demonstrated to modify the ruminal biohydrogenation process and milk fatty acid profile. Measures for using tannins must consider their antinutritional properties (that could cause dry matter intake and digestibility reduction with consequences on productive and reproductive performances, depending however by type (condensed or hydrolysable tannins) and dose [158]. Benchaar et al. [138] reported only a minor effect of Yucca schidigera saponins and no effect of quebracho tree tannins on milk yield and fatty acid profile. Senturk et al. [144] investigated the influence of tannins on protein metabolism and negative energy balance, which resulted in decreased BHB. Wang et al. [140] reported that tea saponins may reduce oxidative stress and improve the immunity system, but the effect depends on the dosage supplementation.
Ruminal protected choline substitute has also raised interest as a feed plant containing herbal choline conjugates due to the antimicrobial and immune stimulating effect of its contained phytoextract. During a three-year study, Gutièrrez et al. [139] revealed that herbal choline inclusion (0.071% of the diet) increased milk yield and fertility, reducing disease (mastitis, abortions) but increasing hypocalcemia disorders.
Research has also been open to Aloe arborescens, which contains polysaccharides and pectins and exhibits anti-inflammatory, immune stimulant, antibacterial, and antioxidant properties [105, 159, 160].
Conclusions
Nutraceuticals provide a valuable tool in feed additives due to their host-protecting functions (antioxidant, anti-inflammatory, antimicrobial, and cell survival effects) to increase productive and reproductive performances. Thus, administration of peripartum nutraceuticals– particularly those discussed in the present review – has drawn attention to their potential health benefits and metabolic responses. The latter opens a wide perspective to further understand the site and mode of action of these compounds towards the main organs primarily involved in the homeorhetic adaptation during the transition period, such as the gastrointestinal epithelia, liver, adipose tissue, rumen, immune system, and uterus. Even more, the data support the use of supplemental nutraceuticals in the transition period to enhance the metabolic, immune, and antioxidant system response and to reduce the release of “signals” responsible for inflammation, immune dysregulation, and metabolic adaptation impairment immediately after parturition. These mechanisms should be further investigated in-depth through a system biology approach, combining performance data with -omics techniques, such as transcriptomics, proteomics, and metabolomics. In vitro studies are also notably encouraged, whereby researchers could target specific hypotheses and successfully answer them at least at the molecular level. However, nutraceuticals are a large class of compounds, and thus, their efficacy is affected by many factors. Several of these include the source, the technique used for production, concentration of the compound, along with the physical condition, diet, rumen pH, animal physiology, interference among compounds and nutrients, and synergistic or antagonistic effects. For these reasons, more studies should be performed to assess the efficacy and toxicity of these natural and potent compounds as well as their role in reducing the need for antibiotics.
Availability of data and materials
Not applicable.
References
Ingvartsen KL, Moyes K. Nutrition, immune function and health of dairy cattle. Animal. 2013;7:112–22 Available from: https://www.cambridge.org/core/product/identifier/S175173111200170X/type/journal_article.
Trevisi E, Minuti A. Assessment of the innate immune response in the periparturient cow. Res Vet Sci. 2018. 47–54.
Minuti A, Bionaz M, Lopreiato V, Janovick NA, Rodriguez-Zas SL, Drackley JK, et al. Prepartum dietary energy intake alters adipose tissue transcriptome profiles during the periparturient period in Holstein dairy cows. J Anim Sci Biotechnol. 2020;11:1. https://doi.org/10.1186/s40104-019-0409-7.
Bradford BJ, Swartz TH. Review: Following the smoke signals: inflammatory signaling in metabolic homeostasis and homeorhesis in dairy cattle. Animal. 2020;14:s144–54 Available from: https://www.cambridge.org/core/product/identifier/S1751731119003203/type/journal_article.
Dormán G, Flachner B, Hajdú I, András CD. Target Identification and Polypharmacology of Nutraceuticals. Nutraceuticals. 2016;263–86. Available from: https://linkinghub.elsevier.com/retrieve/pii/B9780128021477000218. Elsevier.
Spears JW, Weiss WP. Role of antioxidants and trace elements in health and immunity of transition dairy cows. Vet J. 2008;176(1):70-6.
Drackley JK. Biology of dairy cows during the transition period: the final frontier? J Dairy Sci. 1999;82:2259–73.
Contreras GA, Strieder-Barboza C, Raphael W. Adipose tissue lipolysis and remodeling during the transition period of dairy cows. J. Anim. Sci. Biotechnol. 2017;8:41.
Drackley JK. ADSA Foundation scholar award. Biology of dairy cows during the transition period: the final frontier? J Dairy Sci. 1999;82:2259–73.
Ingvartsen KL, Moyes K, Ingvartsen, K.L. And KM. Nutrition, immune function and health of dairy cattle. Page in animal. Animal. 2013;7:112–122.
Kvidera SK, Horst EA, Abuajamieh M, Mayorga EJ, Fernandez MVS, Baumgard LH. Glucose requirements of an activated immune system in lactating Holstein cows. J Dairy Sci. 2017;100:2360–74.
Herdt TH. Ruminant adaptation to negative energy balance. Influences on the etiology of ketosis and fatty liver. Vet Clin North Am Food Anim Pract. 2000;16(2):215-30.
Bertoni G, Trevisi E. Use of the liver activity index and other metabolic variables in the assessment of metabolic health in dairy herds. Vet Clin North Am- Food Anim Pract. 2013. 413–31.
Reis WL, Yi C-X, Gao Y, Tschöp MH, Stern JE. Brain innate immunity regulates hypothalamic Arcuate neuronal activity and feeding behavior. Endocrinology. 2015;156:1303–15 Available from: https://academic.oup.com/endo/article-lookup/doi/10.1210/en.2014-1849.
Sartin JL, Daniel JA, Whitlock BK, Wilborn RR. Selected hormonal and neurotransmitter mechanisms regulating feed intake in sheep. Animal. 2010;4:1781–1789. Available from: http://www.journals.cambridge.org/abstract_S1751731110001497.
Minuti A, Palladino A, Khan MJ, Alqarni S, Agrawal A, Piccioli-Capelli F, et al. Abundance of ruminal bacteria, epithelial gene expression, and systemic biomarkers of metabolism and inflammation are altered during the peripartal period in dairy cows. J Dairy Sci. 2015;98:8940–51 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030215006918.
Steele MA, Schiestel C, AlZahal O, Dionissopoulos L, Laarman AH, Matthews JC, et al. The periparturient period is associated with structural and transcriptomic adaptations of rumen papillae in dairy cattle. J Dairy Sci. 2015;98:2583–95 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030215000892.
Douglas GN, Overton TR, Bateman HG, Dann HM, Drackley JK. Prepartal plane of nutrition, regardless of dietary energy source, affects periparturient metabolism and dry matter intake in Holstein cows. J Dairy Sci. 2006;89(6):2141-57.
Sordillo LM, Contreras GA, Aitken SL. Metabolic factors affecting the inflammatory response of periparturient dairy cows. Anim Heal Res Rev Cambridge Univ Press. 2009;10:53–63.
Douglas GN, Rehage J, Beaulieu AD, Bahaa AO, Drackley JK. Prepartum nutrition alters fatty acid composition in plasma, adipose tissue, and liver lipids of Periparturient dairy cows. J Dairy Sci. 2007;90:2941–59 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030207701066.
Moallem U. Invited review: roles of dietary n-3 fatty acids in performance, milk fat composition, and reproductive and immune systems in dairy cattle. J Dairy Sci. 2018;101:8641–61 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030218307318.
Petit HV, Germiquet C, Lebel D. Effect of feeding whole, unprocessed sunflower seeds and flaxseed on milk production, milk composition, and prostaglandin secretion in dairy cows. J Dairy Sci. 2004;87:3889–98. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030204735286.
Petit HV, Ivan M, Mir PS. Effects of flaxseed on protein requirements and N excretion of dairy cows fed diets with two protein concentrations. J Dairy Sci. 2005;88:1755–64. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030205728502.
Mustafa A, Chouinard P, Christensen D. Effects of feeding micronised flaxseed on yield and composition of milk from Holstein cows. J Sci Food Agric. 2003;83:920–6 Available from: http://doi.wiley.com/10.1002/jsfa.1430.
Akraim F, Nicot MC, Juaneda P, Enjalbert F. Conjugated linolenic acid (CLnA), conjugated linoleic acid (CLA) and other biohydrogenation intermediates in plasma and milk fat of cows fed raw or extruded linseed. Animal. 2007;1:835–43 Available from: https://www.cambridge.org/core/product/identifier/S175173110700002X/type/journal_article.
Zachut M, Arieli A, Lehrer H, Livshitz L, Yakoby S, Moallem U. Effects of increased supplementation of n-3 fatty acids to transition dairy cows on performance and fatty acid profile in plasma, adipose tissue, and milk fat. J Dairy Sci. 2010;93:5877–89 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030210006399.
Moallem U. The effects of extruded flaxseed supplementation to high-yielding dairy cows on milk production and milk fatty acid composition. Anim Feed Sci Technol. 2009;152:232–42 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0377840109001382.
Neveu C, Baurhoo B, Mustafa A. Effect of feeding extruded flaxseed with different forage:concentrate ratios on the performance of dairy cows. J Dairy Sci. 2013;96:3886–94 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030213002981.
Gonthier C, Mustafa AF, Ouellet DR, Chouinard PY, Berthiaume R, Petit HV. Feeding micronized and extruded flaxseed to dairy cows: effects on blood parameters and milk fatty acid composition. J Dairy Sci. 2005;88(2):748-56.
Suksombat W, Thanh LP, Meeprom C, Mirattanaphrai R. Effect of linseed oil supplementation on performance and milk fatty acid composition in dairy cows. Anim Sci J. 2016;87:1545–53 Available from: http://doi.wiley.com/10.1111/asj.12609.
Moallem U, Shafran A, Zachut M, Dekel I, Portnick Y, Arieli A. Dietary α-linolenic acid from flaxseed oil improved folliculogenesis and IVF performance in dairy cows, similar to eicosapentaenoic and docosahexaenoic acids from fish oil. Reproduction. 2013;146:603–14 Available from: https://rep.bioscientifica.com/view/journals/rep/146/6/603.xml.
Pirondini M, Colombini S, Mele M, Malagutti L, Rapetti L, Galassi G, et al. Effect of dietary starch concentration and fish oil supplementation on milk yield and composition, diet digestibility, and methane emissions in lactating dairy cows. J Dairy Sci. 2015;98:357–72 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030214007681.
AbuGhazaleh AA, Schingoethe DJ, Hippen AR, Kalscheur KF, Whitlock LA. Fatty acid profiles of milk and rumen digesta from cows fed fish oil, extruded soybeans or their blend. J Dairy Sci. 2002;85:2266–76. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030202743063.
Shingfield KJ, Griinari JM. Role of biohydrogenation intermediates in milk fat depression. Eur J Lipid Sci Technol. 2007;109:799–816 Available from: http://doi.wiley.com/10.1002/ejlt.200700026.
Calder PC. The relationship between the fatty acid composition of immune cells and their function. Prostaglandins Leukot Essent Fat Acids. 2008;79(3-5):101-8.
Sordillo LM, Raphael W. Significance of metabolic stress, lipid mobilization, and inflammation on transition cow disorders. Vet. Clin. North Am. - Food Anim. Pract. 2013; 267–78.
Brassard P, Larbi A, Grenier A, Frisch F, Fortin C, Carpentier AC, et al. Modulation of T-cell signalling by non-esterified fatty acids. Prostaglandins Leukot Essent Fat Acids. 2007;77(5-6):337-43.
Lee JY, Plakidas A, Lee WH, Heikkinen A, Chanmugam P, Bray G, et al. Differential modulation of toll-like receptors by fatty acids: preferential inhibition by n-3 polyunsaturated fatty acids. J Lipid Res. 2003;44:479–86.
Lessard M, Gagnon N, Godson DL, Petit HV. Influence of parturition and diets enriched in n-3 or n-6 polyunsaturated fatty acids on immune response of dairy cows during the transition period. J Dairy Sci. 2004;87:2197–210.
Lee JY, Zhao L. Hwang DH. Nutr Rev: Modulation of pattern recognition receptor-mediated inflammation and risk of chronic diseases by dietary fatty acids. 2010:68(1):38-61.
Scalia D, Lacetera N, Bernabucci U, Demeyere K, Duchateau L, Burvenich C. In vitro effects of nonesterified fatty acids on bovine neutrophils oxidative burst and viability. J Dairy Sci. 2006;89(1):147-54.
Kawasaki T, Kawai T. Toll-like receptor signaling pathways. Front Immunol. 2014;5:461.
Greco LF, Neves Neto JT, Pedrico A, Lima FS, Bisinotto RS, Martinez N, et al. Effects of altering the ratio of dietary n-6 to n-3 fatty acids on spontaneous luteolysis in lactating dairy cows. J Dairy Sci. 2018;101:10536–56.
Barquissau V, Ghandour RA, Ailhaud G, Klingenspor M, Langin D, Amri EZ, et al. Control of adipogenesis by oxylipins, GPCRs and PPARs. Biochimie. 2017;136:3-11.
Lee JY, Zhao L, Youn HS, Weatherill AR, Tapping R, Feng L, et al. Saturated fatty acid activates but polyunsaturated fatty acid inhibits toll-like receptor 2 dimerized with toll-like receptor 6 or 1. J Biol Chem. 2004;279:16971–9.
Contreras GA, Raphael W, Mattmiller SA, Gandy JC, Sordillo LM. Nonesterified fatty acids modify inflammatory response and eicosanoid biosynthesis in bovine endothelial cells. J Dairy Sci. 2012;95:5011–23.
Silvestre FT, Carvalho TSM, Francisco N, Santos JEP, Staples CR, Jenkins TC, et al. Effects of differential supplementation of fatty acids during the peripartum and breeding periods of Holstein cows: I. Uterine and metabolic responses, reproduction, and lactation. J Dairy Sci. 2011;94:189–204 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030210006879.
Ballou MA, Gomes RC, DePeters EJ. Supplemental fish oil does not alter immune competence or the pathophysiological response to an intramammary infusion of endotoxin in peri-partum multiparous Holstein cows. J Dairy Res. 2009;76:165–72 Available from: https://www.cambridge.org/core/product/identifier/S0022029908003804/type/journal_article.
Kushibiki S, Hodate K, Shingu H, Ueda Y, Shinoda M, Mori Y, et al. Insulin resistance induced in dairy steers by tumor necrosis factor alpha is partially reversed by 2,4–thiazolidinedione. Domest Anim Endocrinol. 2001;21:25–37.
Ballou MA, Gomes RC, Juchem SO, DePeters EJ. Effects of dietary supplemental fish oil during the peripartum period on blood metabolites and hepatic fatty acid compositions and total triacylglycerol concentrations of multiparous Holstein cows. J Dairy Sci. 2009;92:657–69.
Perdomo MC, Santos JE, Badinga L. Trans-10, cis-12 conjugated linoleic acid and the PPAR-γ agonist rosiglitazone attenuate lipopolysaccharide-induced TNF-α production by bovine immune cells. Domest Anim Endocrinol. 2011;41:118–25.
Saremi B, Al-Dawood A, Winand S, Müller U, Pappritz J, von Soosten D, et al. Bovine haptoglobin as an adipokine: Serum concentrations and tissue expression in dairy cows receiving a conjugated linoleic acids supplement throughout lactation. Vet Immunol Immunopathol. 2012;146:201–11 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0165242712000840.
Silvestre FT, Carvalho TSM, Crawford PC, Santos JEP, Staples CR, Jenkins T, et al. Effects of differential supplementation of fatty acids during the peripartum and breeding periods of Holstein cows: II. Neutrophil fatty acids and function, and acute phase proteins. J Dairy Sci. 2011;94:2285–301.
Cheng W-L, Lii C-K, Chen H-W, Lin T-H, Liu K-L. Contribution of conjugated linoleic acid to the suppression of inflammatory responses through the regulation of the NF-κB pathway. J Agric Food Chem. 2004;52:71–8. Available from: https://pubs.acs.org/doi/10.1021/jf0348626.
Petit HV, Benchaar C. Milk production, milk composition, blood composition, and conception rate of transition dairy cows fed different profiles of fatty acids. Can J Anim Sci. 2007;87:591–600 Available from: http://www.nrcresearchpress.com/doi/10.4141/CJAS07027.
Koletzko B, Larqué E, Demmelmair H. Placental transfer of long-chain polyunsaturated fatty acids (LC-PUFA). J Perinat Med. 2007;35(S1):S5-11.
Innis SM. Dietary omega 3 fatty acids and the developing brain. Brain Res. 2008;1237:35–43.
Stoffel W, Holz B, Jenke B, Binczek E, Günter RH, Kiss C, et al. Δ6-Desaturase (FADS2) deficiency unveils the role of ω3- and ω6-polyunsaturated fatty acids. EMBO J. 2008;27:2281–92.
Ponter AA, Parsy A-E, Saadé M, Mialot J-P, Ficheux C, Duvaux-Ponter C, et al. Effect of a supplement rich in linolenic acid added to the diet of post partum dairy cows on ovarian follicle growth, and milk and plasma fatty acid compositions. Reprod Nutr Dev. 2006;46:19–29 Available from: http://www.edpsciences.org/10.1051/rnd:2005058.
Hochi S, Kimura K, Hanada A. Effect of linoleic acid-albumin in the culture medium on freezing sensitivity of in vitro-produced bovine morulae. Theriogenology. 1999;52:497–504 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0093691X99001466.
Leroy JLMR, Vanholder T, Mateusen B, Christophe A, Opsomer G, de Kruif A, et al. Non-esterified fatty acids in follicular fluid of dairy cows and their effect on developmental capacity of bovine oocytes in vitro. Reproduction. 2005;130:485–95 Available from: https://rep.bioscientifica.com/view/journals/rep/130/4/1300485.xml.
Zachut M, Dekel I, Lehrer H, Arieli A, Arav A, Livshitz L, et al. Effects of dietary fats differing in n-6:n-3 ratio fed to high-yielding dairy cows on fatty acid composition of ovarian compartments, follicular status, and oocyte quality. J Dairy Sci. 2010;93:529–45 Available from: https://linkinghub.elsevier.com/retrieve/pii/S002203021071496X.
Sinedino LDP, Honda PM, Souza LRL, Lock AL, Boland MP, Staples CR, et al. Effects of supplementation with docosahexaenoic acid on reproduction of dairy cows. Reproduction. 2017;153:707–23 Available from: https://rep.bioscientifica.com/view/journals/rep/153/5/707.xml.
Mattos R, Staples CR, Arteche A, Wiltbank MC, Diaz FJ, Jenkins TC, et al. The effects of feeding fish oil on uterine secretion of PGF2α, milk composition, and metabolic status of periparturient Holstein cows. J Dairy Sci. 2004;87:921–32.
Trebble TM, Wootton SA, Miles EA, Mullee M, Arden NK, Ballinger AB, et al. Prostaglandin E2 production and T cell function after fish-oil supplementation: response to antioxidant cosupplementation. Am J Clin Nutr. 2003;78:376–82.
Fouladi-Nashta AA, Wonnacott KE, Gutierrez CG, Gong JG, Sinclair KD, Garnsworthy PC, et al. Oocyte quality in lactating dairy cows fed on high levels of n-3 and n-6 fatty acids. Reproduction. 2009;138:771–81.
Bender K, Walsh S, Evans ACO, Fair T, Brennan L. Metabolite concentrations in follicular fluid may explain differences in fertility between heifers and lactating cows. Reproduction. 2010;139:1047–55.
Otto JR, Freeman MJ, Malau-Aduli BS, Nichols PD, Lane PA, Malau-Aduli AEO. Reproduction and fertility parameters of dairy cows supplemented with Omega-3 fatty acid-rich canola oil. Annu Res Rev Biol. 2014;4:1611–36.
Kim DH, Lee HJ, Amanullah SM, Adesogan AT, Kim SC. Effects of dietary n-6/n-3 fatty acid ratio on nutrient digestibility and blood metabolites of Hanwoo heifers. Anim Sci J. 2016;87:46–53.
Stocco DM, Clark BJ. Regulation of the acute production of steroids in Steroidogenic cells. Endocr Rev. 1996;17:221–44.
Wang X, Walsh LP, Reinhart AJ, Stocco DM. The role of Arachidonic acid in Steroidogenesis and Steroidogenic acute regulatory (StAR) gene and Protein expression. J Biol Chem. 2000;275:20204–9.
Zeisel SH. Choline: an important nutrient in brain development, liver function and carcinogenesis. J Am Coll Nutr. 1992;11:473–81.
McFadden JW, Girard CL, Tao S, Zhou Z, Bernard JK, Duplessis M, et al. Symposium review: one-carbon metabolism and methyl donor nutrition in the dairy cow. J Dairy Sci. 2020;103(6):5668-83.
Pinotti L, Baldi A, Dell’Orto V. Comparative mammalian choline metabolism with emphasis on the high-yielding dairy cow. Nutr Res Rev. 2002;15:315–32.
Neill AR, Grime DW, Dawson RMC. Conversion of choline methyl groups through trimethylamine into methane in the rumen. Biochem J. 1978;170:529–35.
Pinotti L, Campagnoli A, Dell’Orto V, Baldi A. Choline: is there a need in the lactating dairy cow? Livest Prod Sci. 2005;98:149–52.
Elek P, Newbold JR, Gaal T, Wagner L, Husveth F. Effects of rumen-protected choline supplementation on milk production and choline supply of periparturient dairy cows. Animal. 2008;2:1595–601.
Zenobi MG, Gardinal R, Zuniga JE, Dias ALG, Nelson CD, Driver JP, et al. Effects of supplementation with ruminally protected choline on performance of multiparous Holstein cows did not depend upon prepartum caloric intake. J Dairy Sci. 2018;101:1088–110 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030217311189.
Zom RLG, van Baal J, Goselink RMA, Bakker JA, de Veth MJ, van Vuuren AM. Effect of rumen-protected choline on performance, blood metabolites, and hepatic triacylglycerols of periparturient dairy cattle. J Dairy Sci. 2011;94:4016–27.
Lima FS, Sá Filho MF, Greco LF, Santos JEP. Effects of feeding rumen-protected choline on incidence of diseases and reproduction of dairy cows. Vet J. 2012;193:140–5.
Batistel F, Arroyo JM, Bellingeri A, Wang L, Saremi B, Parys C, et al. Ethyl-cellulose rumen-protected methionine enhances performance during the periparturient period and early lactation in Holstein dairy cows. J Dairy Sci. 2017;100:7455–67.
Osorio JS, Trevisi E, Ji P, Drackley JK, Luchini D, Bertoni G, et al. Biomarkers of inflammation, metabolism, and oxidative stress in blood, liver, and milk reveal a better immunometabolic status in peripartal cows supplemented with Smartamine M or MetaSmart. J Dairy Sci. 2014;97:7437–50.
Zhou Z, Vailati-Riboni M, Trevisi E, Drackley JK, Luchini DN, Loor JJ. Better postpartal performance in dairy cows supplemented with rumen-protected methionine compared with choline during the peripartal period. J Dairy Sci. 2016;99:8716–32 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030216306245.
Alharthi AS, Batistel F, Abdelmegeid MK, Lascano G, Parys C, Helmbrecht A, et al. Maternal supply of methionine during late-pregnancy enhances rate of Holstein calf development in utero and postnatal growth to a greater extent than colostrum source. J Anim Sci Biotechnol. 2018;9:83.
Coleman DN, Alharthi A, Lopreiato V, Trevisi E, Miura M, Pan Y-X, et al. Choline supply during negative nutrient balance alters hepatic cystathionine β-synthase, intermediates of the methionine cycle and transsulfuration pathway, and liver function in Holstein cows. J Dairy Sci. 2019;102:8319–31 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030219304114.
Zhou Z, Bulgari O, Vailati-Riboni M, Trevisi E, Ballou MA, Cardoso FC, et al. Rumen-protected methionine compared with rumen-protected choline improves immunometabolic status in dairy cows during the peripartal period. J Dairy Sci. 2016;99:8956–69 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030216305835.
Goselink RMA, van Baal J, Widjaja HCA, Dekker RA, Zom RLG, de Veth MJ, et al. Effect of rumen-protected choline supplementation on liver and adipose gene expression during the transition period in dairy cattle. J Dairy Sci. 2013;96:1102–16.
Pinotti L, Baldi A, Politis I, Rebucci R, Sangalli L, Dell’Orto V. Rumen-protected choline administration to transition cows: effects on Milk production and vitamin E status. J Vet Med Ser A. 2003;50:18–21.
Leiva T, Cooke RF, Brandão AP, Marques RS, Vasconcelos JLM. Effects of rumen-protected choline supplementation on metabolic and performance responses of transition dairy cows1. J Anim Sci. 2015;93:1896–904.
Zhou Z, Ferdous F, Montagner P, Luchini DN, Corrêa MN, Loor JJ. Methionine and choline supply during the peripartal period alter polymorphonuclear leukocyte immune response and immunometabolic gene expression in Holstein cows. J Dairy Sci. 2018;101:10374–82 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030218307975.
Vailati-Riboni M, Zhou Z, Jacometo CB, Minuti A, Trevisi E, Luchini DN, et al. Supplementation with rumen-protected methionine or choline during the transition period influences whole-blood immune response in periparturient dairy cows. J Dairy Sci. 2017;100:3958–68 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030217302412.
Batistel F, Arroyo JM, Garces CIM, Trevisi E, Parys C, Ballou MA, et al. Ethyl-cellulose rumen-protected methionine alleviates inflammation and oxidative stress and improves neutrophil function during the periparturient period and early lactation in Holstein dairy cows. J Dairy Sci. 2017;101:480–90.
Alharthi AS, Coleman DN, Liang Y, Batistel F, Elolimy AA, Yambao RC, et al. Hepatic 1-carbon metabolism enzyme activity, intermediate metabolites, and growth in neonatal Holstein dairy calves are altered by maternal supply of methionine during late pregnancy. J Dairy Sci. 2019;102:10291–303.
Jacometo CB, Zhou Z, Luchini D, Trevisi E, Corrêa MN, Loor JJ. Maternal rumen-protected methionine supplementation and its effect on blood and liver biomarkers of energy metabolism, inflammation, and oxidative stress in neonatal Holstein calves. J Dairy Sci. 2016;99:6753–63 Available from: http://www.ncbi.nlm.nih.gov/pubmed/27209133.
Gruffat D, Durand D, Graulet B, Bauchart D. Regulation of VLDL synthesis and secretion in the liver. Reprod Nutr Dev. 1996;36:375–89.
Cadórniga-Valiño C, Grummer RR, Armentano LE, Donkin SS, Bertics SJ. Effects of fatty acids and hormones on fatty acid metabolism and gluconeogenesis in bovine hepatocytes. J Dairy Sci. 1997;80:646–56.
Schwab CG, Bozak CK, Whitehouse NL, Mesbah MMA. Amino acid limitation and flow to duodenum at four stages of lactation. 1. Sequence of lysine and methionine limitation. J Dairy Sci. 1992;75:3486–502.
Brosnan JT, Brosnan ME. The sulfur-containing amino acids: an overview. J Nutr. 2006;136:1636S–40S.
Emmanuel B, Kennelly JJ. Kinetics of methionine and choline and their incorporation into plasma lipids and Milk components in lactating goats. J Dairy Sci. 1984;67:1912–8.
National Research Council. Nutrient Requirements of Dairy Cattle. 7th ed. Nutr. Requir. Dairy Cattle. Washington, D.C.: National Academies Press (2001); 2001. Available from: http://www.nap.edu/catalog/9825.
Dalbach KF, Larsen M, Raun BML, Kristensen NB. Effects of supplementation with 2-hydroxy-4-(methylthio)-butanoic acid isopropyl ester on splanchnic amino acid metabolism and essential amino acid mobilization in postpartum transition Holstein cows. J Dairy Sci. 2011;94:3913–27.
Zhou Z, Vailati-Riboni M, Luchini D, Loor J. Methionine and choline supply during the periparturient period alter plasma amino acid and one-carbon metabolism profiles to various extents: potential role in hepatic metabolism and antioxidant status. Nutrients. 2016;9:10. Available from: http://www.mdpi.com/2072-6643/9/1/10.
Lopreiato V, Vailati-Riboni M, Bellingeri A, Khan I, Farina G, Parys C, et al. Inflammation and oxidative stress transcription profiles due to in vitro supply of methionine with or without choline in unstimulated blood polymorphonuclear leukocytes from lactating Holstein cows. J Dairy Sci. 2019;102:10395–410 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030219307209.
Jacometo CB, Osorio JS, Socha M, Corrêa MN, Piccioli-Cappelli F, Trevisi E, et al. Maternal consumption of organic trace minerals alters calf systemic and neutrophil mRNA and microRNA indicators of inflammation and oxidative stress. J Dairy Sci. 2015;98:7717–29 Available from: http://linkinghub.elsevier.com/retrieve/pii/S0022030215006128.
Trevisi E, Zecconi A, Cogrossi S, Razzuoli E, Grossi P, Amadori M. Strategies for reduced antibiotic usage in dairy cattle farms. Res Vet Sci. 2014;96:229–33 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0034528814000253.
Newbold CJ, Wallace RJ, Mcintosh FM. Mode of action of the yeast Saccharomyces cerevisiae as a feed additive for ruminants. Br J Nutr. 1996;76:249–61. Available from: https://www.cambridge.org/core/product/identifier/S0007114596001328/type/journal_article.
Uyeno Y, Akiyama K, Hasunuma T, Yamamoto H, Yokokawa H, Yamaguchi T, et al. Effects of supplementing an active dry yeast product on rumen microbial community composition and on subsequent rumen fermentation of lactating cows in the mid-to-late lactation period. Anim Sci J. 2017;88:119–24 Available from: http://doi.wiley.com/10.1111/asj.12612.
Pinloche E, McEwan N, Marden J-P, Bayourthe C, Auclair E, Newbold CJ. The effects of a probiotic yeast on the bacterial diversity and population structure in the rumen of cattle. White BA, editor. PLoS One. 2013;8:e67824. Available from: http://dx.plos.org/10.1371/journal.pone.0067824.
Al Ibrahim RM, Kelly AK, O’Grady L, Gath VP, McCarney C, Mulligan FJ. The effect of body condition score at calving and supplementation with Saccharomyces cerevisiae on milk production, metabolic status, and rumen fermentation of dairy cows in early lactation. J Dairy Sci. 2010;93:5318–28. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030210005746.
Nocek JE, Kautz WP. Direct-Fed Microbial Supplementation on Ruminal Digestion, Health, and Performance of Pre- and Postpartum Dairy Cattle. J Dairy Sci. 2006;89:260–6 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030206720902.
Dann HM, Drackley JK, McCoy GC, Hutjens MF, Garrett JE. Effects of yeast culture (Saccharomyces cerevisiae) on prepartum intake and postpartum intake and milk production of Jersey cows. J Dairy Sci. 2000;83:123–7. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030200748636.
Kim HS, Ahn BS, Chung SG, Moon YH, Ha JK, Seo IJ, et al. Effect of yeast culture, fungal fermentation extract and non-ionic surfactant on performance of Holstein cows during transition period. Anim Feed Sci Technol. 2006;126:23–9 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0377840105002634.
Nocek JE, Holt MG, Oppy J. Effects of supplementation with yeast culture and enzymatically hydrolyzed yeast on performance of early lactation dairy cattle. J Dairy Sci. 2011;94:4046–56 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030211004085.
Olagaray KE, Sivinski SE, Saylor BA, Mamedova LK, Sauls-Hiesterman JA, Yoon I, et al. Effect of Saccharomyces cerevisiae fermentation product on feed intake parameters, lactation performance, and metabolism of transition dairy cattle. J Dairy Sci. 2019;102:8092–107. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030219306186.
Knoblock CE, Shi W, Yoon I, Oba M. Effects of supplementing a Saccharomyces cerevisiae fermentation product during the periparturient period on the immune response of dairy cows fed fresh diets differing in starch content. J Dairy Sci. 2019;102:6199–209. Available from: https://linkinghub.elsevier.com/retrieve/pii/S002203021930390X.
Zaworski EM, Shriver-Munsch CM, Fadden NA, Sanchez WK, Yoon I, Bobe G. Effects of feeding various dosages of Saccharomyces cerevisiae fermentation product in transition dairy cows. J Dairy Sci. 2014;97:3081–98. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030214001738.
Ramsing EM, Davidson JA, French PD, Yoon I, Keller M, Peters-Fleckenstein H. Effects of yeast culture on peripartum intake and milk production of primiparous and multiparous Holstein cows. Prof Anim Sci. 2009;25:487–95 Available from: http://linkinghub.elsevier.com/retrieve/pii/S1080744615307397.
Robinson PH. Effect of yeast culture (Saccharomyces cerevisiae) on adaptation of cows to diets postpartum. J Dairy Sci. 1997;80:1119–25. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030297760387.
Robinson PH, Garrett JE. Effect of yeast culture (Saccharomyces cerevisiae) on adaptation of cows to postpartum diets and on lactational performance. J Anim Sci. 1999;77:988 Available from: https://academic.oup.com/jas/article/77/4/988-999/4625401.
Poppy GD, Rabiee AR, Lean IJ, Sanchez WK, Dorton KL, Morley PS. A meta-analysis of the effects of feeding yeast culture produced by anaerobic fermentation of Saccharomyces cerevisiae on milk production of lactating dairy cows. J Dairy Sci. 2012;95:6027–41. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030212006212.
Shen Y, Wang H, Ran T, Yoon I, Saleem AM, Yang W. Influence of yeast culture and feed antibiotics on ruminal fermentation and site and extent of digestion in beef heifers fed high grain rations1. J Anim Sci. 2018;96:3916–27 Available from: https://academic.oup.com/jas/article/96/9/3916/5060825.
Shi W, Knoblock CE, Murphy KV, Bruinjé TC, Yoon I, Ambrose DJ, et al. Effects of supplementing a Saccharomyces cerevisiae fermentation product during the periparturient period on performance of dairy cows fed fresh diets differing in starch content. J Dairy Sci. 2019;102:3082–96. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030219301225.
Williams PE, Tait CAG, Innes GM, Newbold CJ. Effects of the inclusion of yeast culture (Saccharomyces cerevisiae plus growth medium) in the diet of dairy cows on milk yield and forage degradation and fermentation patterns in the rumen of steers. J Anim Sci. 1991;69:3016–26 Available from: https://academic.oup.com/jas/article/69/7/3016-3026/4631864.
Bach A, Guasch I, Elcoso G, Chaucheyras-Durand F, Castex M, Fàbregas F, et al. Changes in gene expression in the rumen and colon epithelia during the dry period through lactation of dairy cows and effects of live yeast supplementation. J Dairy Sci. 2018;101:2631–40 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030217312055.
Chen Y, Oba M, Guan LL. Variation of bacterial communities and expression of toll-like receptor genes in the rumen of steers differing in susceptibility to subacute ruminal acidosis. Vet Microbiol. 2012;159:451–9 Available from: http://www.ncbi.nlm.nih.gov/pubmed/22622335.
Abreu MT. Toll-like receptor signalling in the intestinal epithelium: how bacterial recognition shapes intestinal function. Nat Rev Immunol. 2010;10:131–44 Available from: http://www.nature.com/articles/nri2707.
Finck DN, Ribeiro FRB, Burdick NC, Parr SL, Carroll JA, Young TR, et al. Yeast supplementation alters the performance and health status of receiving cattle. Prof Anim Sci. 2014;30:333–41. Available from: http://linkinghub.elsevier.com/retrieve/pii/S108074461530125X.
Duff GC, Galyean ML. Board-invited review: recent advances in management of highly stressed, newly received feedlot cattle. J Anim Sci. 2007;85:823–40 Available from: http://www.ncbi.nlm.nih.gov/pubmed/17085724.
Sanchez NCB, Young TR, Carroll JA, Corley JR, Rathmann RJ, Johnson BJ. Yeast cell wall supplementation alters the metabolic responses of crossbred heifers to an endotoxin challenge. Innate Immun. 2014;20:104–12 Available from: http://journals.sagepub.com/doi/10.1177/1753425913482152.
Samuelsen ABC, Schrezenmeir J, Knutsen SH. Effects of orally administered yeast-derived beta-glucans: A review. Mol Nutr Food Res. 2014;58:183–93 Available from: http://doi.wiley.com/10.1002/mnfr.201300338.
Onderdonk AB, Cisneros RL, Hinkson P, Ostroff G. Anti-infective effect of poly-beta 1-6-glucotriosyl-beta 1-3-glucopyranose glucan in vivo. Infect Immun. 1992;60:1642–7 Available from: http://www.ncbi.nlm.nih.gov/pubmed/1548086.
Yuan K, Mendonça LGD, Hulbert LE, Mamedova LK, Muckey MB, Shen Y, et al. Yeast product supplementation modulated humoral and mucosal immunity and uterine inflammatory signals in transition dairy cows. J Dairy Sci. 2015;98:3236–46 Available from: https://linkinghub.elsevier.com/retrieve/pii/S0022030215001277.
Tassoul MD, Shaver RD. Effect of a mixture of supplemental dietary plant essential oils on performance of periparturient and early lactation dairy cows. J Dairy Sci. 2009;92(4):1734-40.
Hammer KA, Carson CF, Riley TV. Antimicrobial activity of essential oils and other plant extracts. J Appl Microbiol. 1999:86(6);985-90.
Wallace RJ. Antimicrobial properties of plant secondary metabolites. Proc Nutr Soc. 2004;63(4):621-9.
Yoruk R, Marshall MR. Physicochemical properties and function of plant polyphenol oxidase: a review. J Food Biochem. 2003;27(5);361-422.
Drong C, Meyer U, von Soosten D, Frahm J, Rehage J, Breves G, et al. Effect of monensin and essential oils on performance and energy metabolism of transition dairy cows. J Anim Physiol Anim Nutr (Berl). 2016;100:537–51.
Benchaar C, Petit HV, Berthiaume R, Whyte TD, Chouinard PY. Effects of addition of essential oils and monensin premix on digestion, ruminal fermentation, milk production, and milk composition in dairy cows. J Dairy Sci. 2006;89:4352–64.
Gutiérrez RA, Gutiérrez A, Sánchez C, Mendoza GD. Effect of including herbal choline in the diet of a dairy herd; a multiyear evaluation. Emirates J Food Agric. 2019;31:477–81.
Wang B, Tu Y, Zhao SP, Hao YH, Liu JX, Liu FH, et al. Effect of tea saponins on milk performance, milk fatty acids, and immune function in dairy cow. J Dairy Sci. 2017;100(10):8043-52.
Braun HS, Schrapers KT, Mahlkow-Nerge K, Stumpff F, Rosendahl J. Dietary supplementation of essential oils in dairy cows: evidence for stimulatory effects on nutrient absorption. Animal. 2019;13(3):518-23.
Oh J, Hristov AN, Lee C, Cassidy T, Heyler K, Varga GA, et al. Immune and production responses of dairy cows to postruminal supplementation with phytonutrients. J Dairy Sci. 2013;96:7830–43.
Yang WZ, Benchaar C, Ametaj BN, Chaves AV, He ML, McAllister TA. Effects of garlic and juniper berry essential oils on ruminal fermentation and on the site and extent of digestion in lactating cows. J Dairy Sci. 2007;90:5671–81.
Şenturk S, Cihan H, Kasap S, Mecitoğlu Z, Temizel M. Effects on negative energy balance of tannin in dairy cattle. Uludağ Üniversitesi Vet Fakültesi Derg. 2015;34:1–7 Available from: http://dergipark.gov.tr/doi/10.30782/uluvfd.385238.
Santos MB, Robinson PH, Williams P, Losa R. Effects of addition of an essential oil complex to the diet of lactating dairy cows on whole tract digestion of nutrients and productive performance. Anim Feed Sci Technol. 2010;157(1-2):64-71.
Benchaar C, Petit HV, Berthiaume R, Ouellet DR, Chiquette J, Chouinardt PY. Effects of essential oils on digestion, ruminai fermentation, rumen microbial populations, milk production, and milk composition in dairy cows fed alfalfa silage or corn silage. J Dairy Sci. 2007;90(2);886-97.
Jouany JP. Optimizing rumen functions in the close-up transition period and early lactation to drive dry matter intake and energy balance in cows. Anim Reprod Sci. 2006;96:250–64.
Humer E, Kröger I, Neubauer V, Schedle K, Reisinger N, Zebeli Q. Supplementing phytogenic compounds or autolyzed yeast modulates ruminal biogenic amines and plasma metabolome in dry cows experiencing subacute ruminal acidosis. J Dairy Sci. 2018;101:9559–74.
Sikkema J, De Bont JAM, Poolman B. Interactions of cyclic hydrocarbons with biological membranes. J Biol Chem. 1994; 269(11):8022-8.
Ultee A, Bennik MHJ, Moezelaar R. The phenolic hydroxyl group of carvacrol is essential for action against the food-borne pathogen Bacillus cereus. Appl Environ Microbiol. 2002; 68(4): 1561–1568.
Feldberg RS, Chang SC, Kotik AN, Nadler M, Neuwirth Z, Sundstrom DC, et al. In vitro mechanism of inhibition of bacterial cell growth by allicin. Antimicrob Agents Chemother. 1988;32(12):1763–8.
Baratta MT, Dorman HJD, Deans SG, Figueiredo AC, Barroso JG, Ruberto G. Antimicrobial and antioxidant properties of some commercial essential oils. Flavour Fragr J. 1998;13(4):235-244.
Smith-Palmer A, Stewart J, Fyfe L. Antimicrobial properties of plant essential oils and essences against five important food-borne pathogens. Lett Appl Microbiol. 1998;26(2):118-22.
Chao SC, Young DG, Oberg CJ. Screening for inhibitory activity of essential oils on selected bacteria, fungi and viruses. J Essent Oil Res. 2000;12(5):639-49.
Griffin SG, Wyllie SG, Markham JL, Leach DN. The role of structure and molecular properties of terpenoids in determining their antimicrobial activity. Flavour Fragr J. 1999;14(5):322-32.
Drong C, Meyer U, von Soosten D, Frahm J, Rehage J, Schirrmeier H, et al. Effects of monensin and essential oils on immunological, haematological and biochemical parameters of cows during the transition period. J Anim Physiol Anim Nutr (Berl). 2017;101(4):791-806.
Liu HW, Zhou DW, Li K. Effects of chestnut tannins on performance and antioxidative status of transition dairy cows. J Dairy Sci. 2013;96(9):5901-7.
Mangan JL. Nutritional effects of tannins in animal feeds. Nutr Res Rev. 1988;1(1):209-31.
Bani P, Grossi P, Lucini L, Pellizzoni M, Minuti A, Trevisi E. Administration of Aloe arborescens homogenate to cattle: interaction with rumen fermentation and gut absorption of aloin. Ital J Anim Sci. 2016;15(2);233-40.
Mezzetti M, Minuti A, Bionaz M, Piccioli-Cappelli F, Trevisi E. Effects of Aloe arborescens Whole Plant Homogenate on Lipid Metabolism, Inflammatory Conditions and Liver Function of Dairy Cows during the Transition Period. Animals. 2020;10:917 Available from: https://www.mdpi.com/2076-2615/10/5/917.
Acknowledgements
The authors gratefully acknowledge support from the “Romeo and Enrica Invernizzi Foundation” (Milan, Italy) and Università Cattolica del Sacro Cuore (Piacenza, Italy).
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
VL, MM, LC, GF, and AM wrote the manuscript. AM and ET supervised the manuscript and accomplished final proofreading. All authors read and approved the final version of the manuscript.
Author information
V. Lopreiato, M. Mezzetti, and G. Ferronato, Post-doc fellow, Department of Animal Sciences, Food and Nutrition, Faculty of Agriculture, Food and Environmental Science, Università Cattolica del Sacro Cuore, Piacenza, 29122, Italy. L. Cattaneo, PhD Student, Department of Animal Sciences, Food and Nutrition, Faculty of Agriculture, Food and Environmental Science, Università Cattolica del Sacro Cuore, Piacenza, 29122, Italy. A. Minuti, Research Fellow, Department of Animal Sciences, Food and Nutrition, Faculty of Agriculture, Food and Environmental Science, Università Cattolica del Sacro Cuore, Piacenza, 29122, Italy. E. Trevisi, Full Professor, Department of Animal Sciences, Food and Nutrition, Faculty of Agriculture, Food and Environmental Science, Università Cattolica del Sacro Cuore, Piacenza, 29122, Italy.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Lopreiato, V., Mezzetti, M., Cattaneo, L. et al. Role of nutraceuticals during the transition period of dairy cows: a review. J Animal Sci Biotechnol 11, 96 (2020). https://doi.org/10.1186/s40104-020-00501-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s40104-020-00501-x